 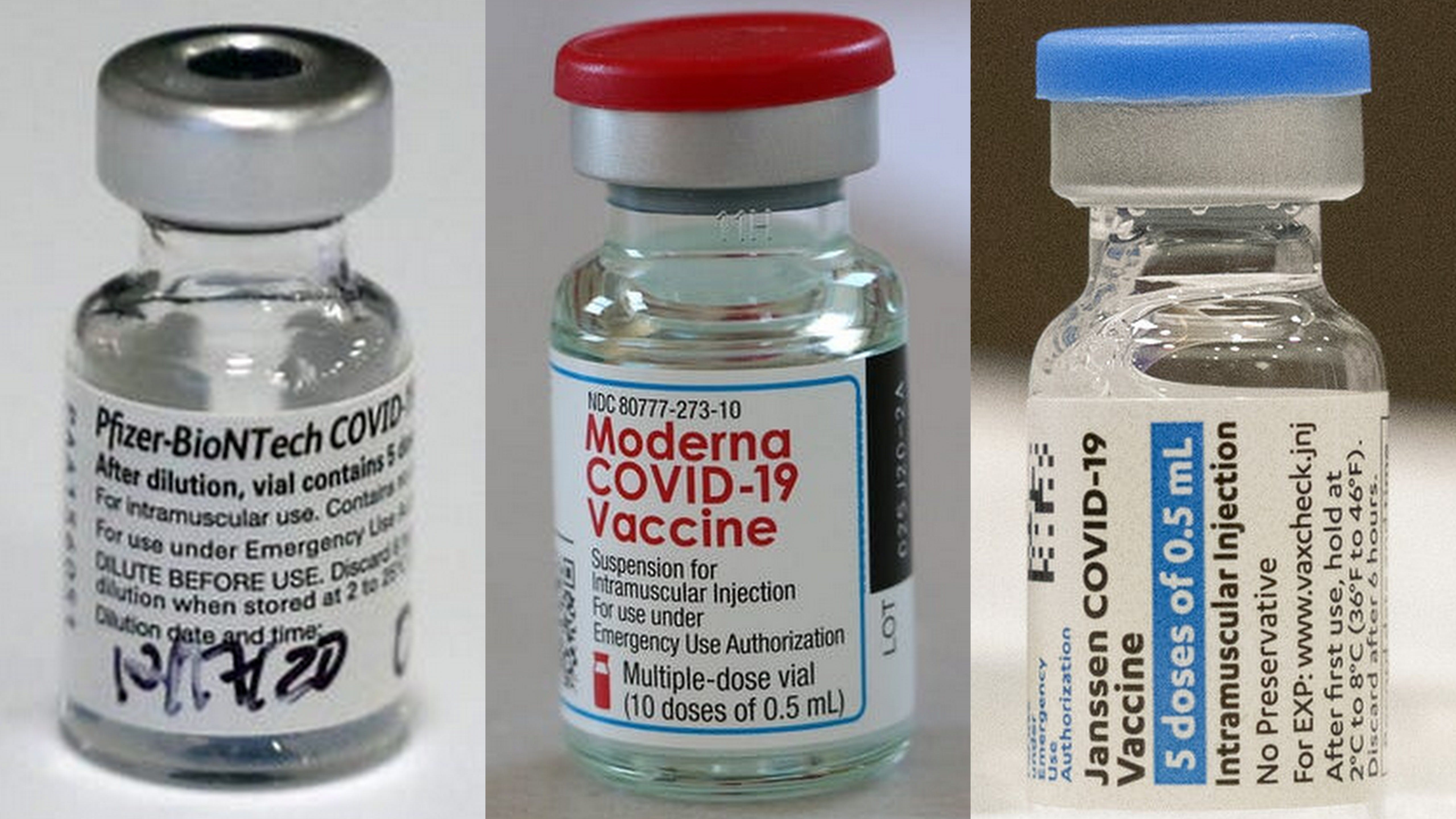
Package leaflets for the COVID-19 vaccines In response, the immune system starts producing antibodies.Ī s used for the basic series of COVID-19 vaccinationsĪ s used for the repeat vaccination against COVID-19 vaccinations If the body encounters the SARS-COV-2 virus, the immune system recognises the spike proteins on the virus. After vaccination, the body responds by producing antibodies against the spike protein. It also contains an adjuvant: a substance that intensifies the body’s immune response to the protein. The vaccine contains very small particles of spike protein, which have been produced in a laboratory. The Novavax vaccine is a protein subunit vaccine.The immune system reacts by producing antibodies, just as it does in response to the mRNA vaccines. The vector vaccine contains an inactivated cold virus (that cannot replicate) with an instruction (code) that tells your cells to make spike proteins. The COVID-19 vaccine made by Janssen is a vector vaccine.The immune system recognises the spike proteins as foreign substances and starts making antibodies. These are the spiky protrusions on the exterior of the coronavirus. These RNA vaccines contain a protective envelope of lipids (fat) with an instruction (code) that tells your cells to make spike proteins. The COVID-19 vaccines made by Pfizer/BioNTech and by Moderna are mRNA vaccines.Uttarakhand officials said the state will import 20 lakh doses of Sputnik vaccine over the next two months.Ĭumulatively, India's vaccination coverage stands at 17.72 Crores as of May 13 which is equivalent to 13.55% (approx) of India's 1.3 billion people.Īmid demands to ramp up domestic supply, Serum Institute and Bharat Biotech have submitted to the Centre their production plan for the next four months, informing that they can scale it up to 10 crore and 7.8 crore doses respectively by August, official sources said.Ĭurently, Bharat Biotech's Covaxin and Oxford-AstraZeneca's Covishield are the only two vaccines are being used in India's inoculation drive against the coronavirus.Read more about these vaccines on Types of COVID-19 vaccines States like Tamil Nadu, Uttar Pradesh, Maharashtra, Karnataka, Andhra Pradesh, Telangana and Delhi have opted for global tenders to meet their needs. 
Besides, other states' governments have floated a tender to import vaccines. Several states in the country have suspended the coronavirus vaccination drive for the 18 to 44 age group as they have run out of stock. Meanwhile, Paul announced that Russia's Sputnik V will be available in markets across the country from early next week. The import license will be granted within 1-2 days". 
He added that "Any vaccine that is approved by FDA and the World Health Organization (WHO) can come to India. Yesterday, US Embassy said that the Biden administration is considering joint production of the Johnson & Johnson Covid-19 vaccine in India by tying up with like Serum Institute of India. Johnson & Johnson did a good job, they accepted this offer under Quad," Paul said on Thursday. "We invite them to manufacture here along with our companies. "I'm hopeful that they'll step forward to increase availability in India," he added.Īccording to Paul, among the three US-based coronavirus vaccine manufacturing companies, only Johnson & Johnson accepted Centre's offer. He added that the Government of India is connected with all three Covid-19 vaccine-making firms. 
Paul underscored that "They (Pfizer and Moderna) had said that they are working in their own way and they would talk of vaccine availability in Q3, in 2021". 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
